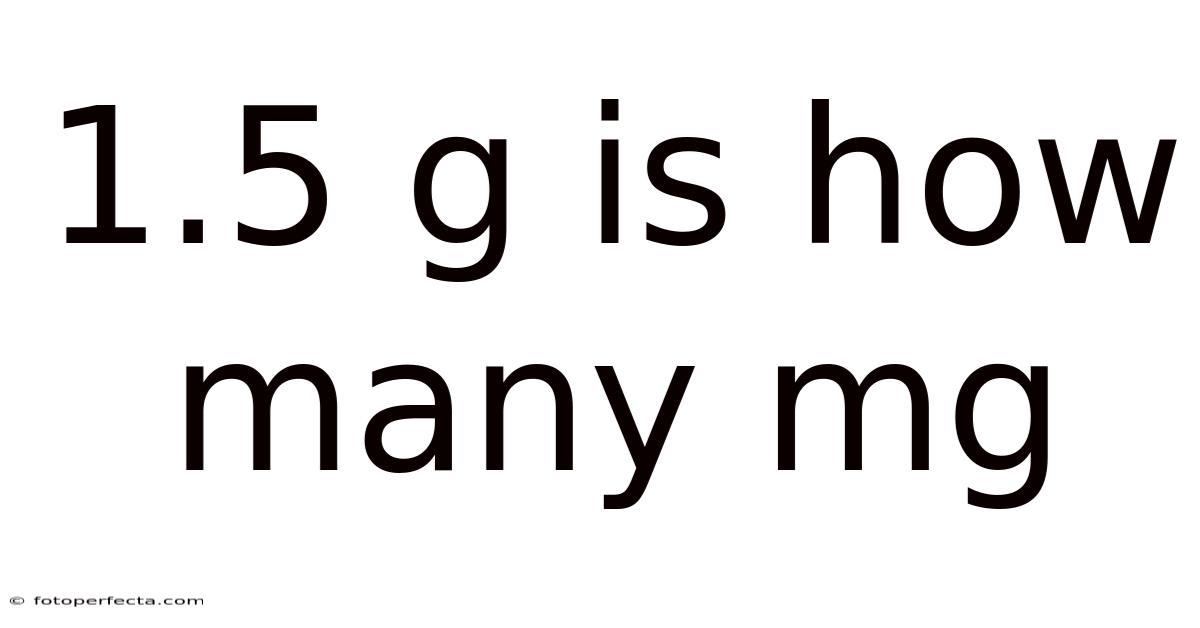1.5 g is how manymg? This question appears simple, yet understanding the conversion between grams and milligrams is essential for anyone working in science, medicine, cooking, or any field that relies on precise measurements. By mastering the relationship between these two metric units, you can confidently interpret labels, follow recipes, administer medication, and conduct experiments without second‑guessing the numbers. In this article we will explore the metric system’s foundation, reveal the exact conversion factor, walk through the calculation for 1.5 grams, provide real‑world examples, highlight common pitfalls, and answer frequently asked questions so you can apply the knowledge with confidence.
Understanding the Metric System
The metric system, also known as the International System of Units (SI), is built on powers of ten, which makes conversions straightforward. The base unit for mass is the kilogram (kg). From there, smaller units are derived by adding prefixes that indicate fractions or multiples of ten:
- kilo‑ (k) = 1 000 × base unit
- hecto‑ (h) = 100 × base unit
- deca‑ (da) = 10 × base unit
- deci‑ (d) = 0.1 × base unit
- centi‑ (c) = 0.01 × base unit - milli‑ (m) = 0.001 × base unit
When we talk about gram (g) and milligram (mg), we are dealing with the gram as the base unit for mass in everyday contexts, and the milli‑ prefix indicating one‑thousandth of a gram. Therefore, the relationship is fixed:
1 gram = 1 000 milligrams
This 1 000‑fold factor is the cornerstone of all gram‑to‑milligram conversions.
Conversion Factor: Grams to Milligrams
Because the metric system scales by tens, converting from grams to milligrams simply requires multiplying the gram value by 1 000. Conversely, to go from milligrams to grams, you divide by 1 000. The formula can be written as:
[\text{milligrams} = \text{grams} \times 1{,}000]
or
[ \text{grams} = \frac{\text{milligrams}}{1{,}000} ]
No complex mathematics is involved; the operation is a simple shift of the decimal point three places to the right when moving from grams to milligrams, and three places to the left when moving in the opposite direction.
Step‑by‑Step Calculation for 1.5 g
Let’s apply the formula to the specific value in question: 1.5 grams.
- Write down the starting amount: 1.5 g
- Multiply by the conversion factor (1 000):
[ 1.5 \times 1{,}000 = 1{,}500 ] - Attach the correct unit: 1 500 mg
Thus, 1.5 g equals 1 500 mg. You can also achieve the same result by moving the decimal point three places to the right: 1.5 → 15 → 150 → 1 500.
Practical Examples Where the Conversion Matters
Medicine and Pharmacology
Drug dosages are frequently expressed in milligrams because the active ingredients are potent even in tiny amounts. A tablet might contain 250 mg of an analgesic, while a liquid suspension could be prescribed at 5 mg per mL. If a doctor orders 1.5 g of a medication, the pharmacist must convert that to 1 500 mg to prepare the correct quantity. Misinterpreting the unit could lead to an overdose or underdose, highlighting why accurate conversion is critical.
Nutrition and Food Labels
Nutrition facts often list nutrients such as sodium, iron, or vitamin C in milligrams per serving. Suppose a breakfast cereal label states that one serving provides 1.5 g of fiber. Converting to milligrams (1 500 mg) lets you compare it directly with other foods that list fiber in mg, facilitating better dietary decisions.
Laboratory Work
In chemistry and biology, reagents are weighed to ensure reactions proceed as expected. A protocol might call for adding 1.5 g of sodium chloride to a solution. The lab balance may display readings in grams, but the stock solution’s concentration could be expressed in mg/mL. Converting the mass to milligrams (1 500 mg) allows you to calculate the volume of stock solution needed with precision.
Jewelry and Precious Metals While gold is usually traded in troy ounces, small-scale jewelry makers sometimes work with grams and milligrams for alloying elements. Knowing that 1.5 g of copper equals 1 500 mg helps them achieve the exact color and hardness they desire in a gold‑copper alloy.
Why Precision MattersThe metric system’s simplicity can lull users into assuming that any approximation is acceptable. However, in fields where safety, efficacy, or reproducibility is paramount, even a small error can have outsized consequences. Consider a medication where the therapeutic window is narrow: a dose of 10 mg might be effective, while 15 mg could cause adverse effects. If a practitioner mistakenly treats 1.5 g as 150 mg (off by a factor of ten), the patient would receive only a tenth of the intended dose, potentially rendering the treatment ineffective. Conversely, confusing milligrams for grams could lead to a tenfold overdose. Therefore, always double‑check the unit before performing any calculation, and when in doubt, write out the conversion step explicitly.
Common Mistakes and How to Avoid Them
| Mistake | Why It Happens | How to Prevent It |
|---|---|---|
| Moving the decimal the wrong direction | Confusing whether to multiply or divide by 1 000 | Remember: grams → milligrams = move decimal right (×1 000); milligrams → grams = move decimal left (÷1 000). |
| Omitting trailing zeros | Writing 1.5 g as 150 mg instead of |
1,500 mg. | A simple oversight that can drastically alter the value. Always include trailing zeros to maintain accuracy. | | Not paying attention to the context | Assuming the unit is implied or doesn’t matter. | Always read the entire problem and understand what is being measured before converting units. | | Relying on mental math | Increasing the chance of errors, especially with complex conversions. | Use a calculator or write out the conversion steps. |
Conclusion
Mastering unit conversions, particularly between grams and milligrams, is a fundamental skill with far-reaching implications. While the metric system aims for simplicity, vigilance and careful attention to detail are crucial to avoid potentially dangerous errors. From ensuring accurate medication dosages to precise laboratory experiments and crafting intricate jewelry, the ability to convert between grams and milligrams is essential. By understanding the principles of conversion, recognizing common mistakes, and employing reliable strategies to avoid them, individuals across various disciplines can confidently navigate the world of measurements and maintain accuracy in their work. Ultimately, the investment in understanding and practicing unit conversions is an investment in precision, safety, and reliable results.
In countless domains, such precision shapes outcomes unimaginable without scrutiny. From engineering specifications to artistic expressions, the fidelity of measurements dictates success. Such considerations extend far beyond individual tasks, influencing global systems and collective progress. By prioritizing accuracy, one cultivates confidence and upholds standards that bind communities together. Mastery here transcends technical skill, becoming a shared responsibility that nourishes trust and cohesion. Ultimately, the pursuit of such clarity ensures that progress remains both reliable and enduring. Thus, maintaining rigor in unit conversion remains a testament to the enduring value of precision in navigating an interconnected world.