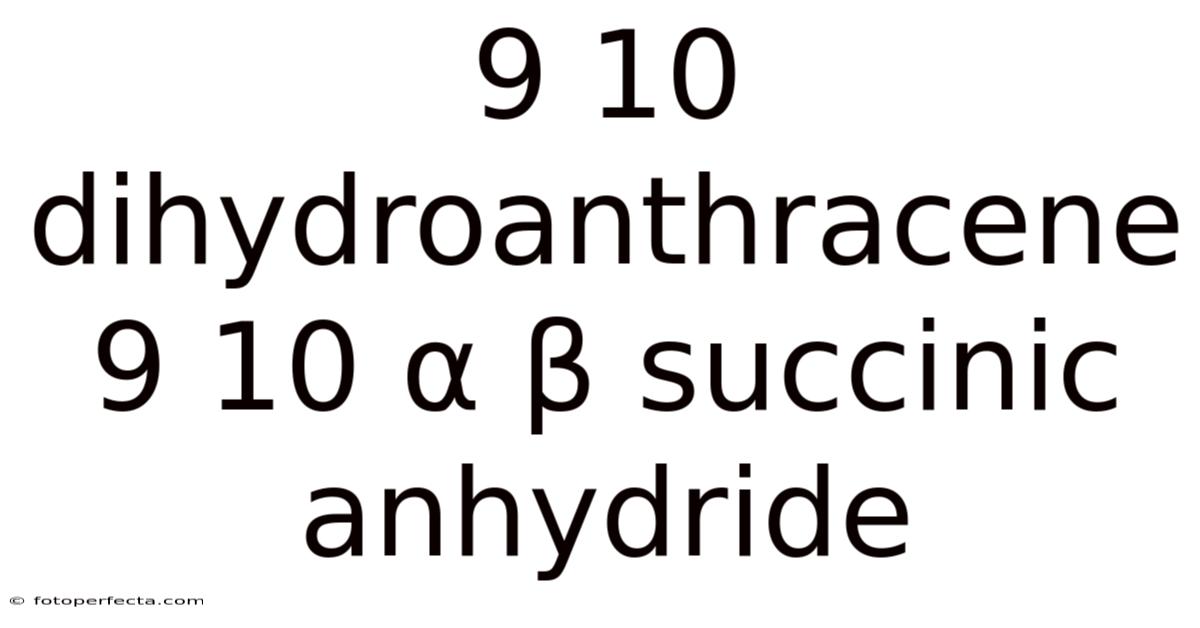
9,10-Dihydroanthracene-9,10-α,β-Succinic Anhydride: Structure, Synthesis, and Applications
Introduction
9,10-Dihydroanthracene-9,10-α,β-succinic anhydride is a specialized organic compound with unique structural and chemical properties. This molecule combines the aromatic framework of anthracene with a succinic anhydride functional group, creating a versatile intermediate in organic synthesis. Its distinctive structure makes it a valuable reagent in reactions requiring controlled reactivity and stability. This article explores its chemical structure, synthesis methods, applications, and safety considerations, providing a comprehensive overview for students and researchers in organic chemistry.
Chemical Structure and Properties
Structural Overview
The compound consists of a 9,10-dihydroanthracene core, where the central benzene ring of anthracene is partially hydrogenated. This saturation at the 9 and 10 positions introduces a non-aromatic, flexible region, enhancing the molecule’s reactivity compared to fully aromatic anthracene. Attached to these positions is a succinic anhydride group, forming a cyclic anhydride bridge between the two carbons. The α and β designations in the name refer to the positions of the oxygen atoms in the anhydride ring relative to the anthracene framework.
The molecular formula is C₁₆H₁₀O₃, with a molecular weight of approximately 254.26 g/mol. The structure can be visualized as follows:
- Anthracene core: Three fused benzene rings with the central ring partially hydrogenated.
- Succinic anhydride group: A five-membered cyclic anhydride (O=C-O-C=O) bridging the 9 and 10 positions.
Physical and Chemical Properties
- Appearance: Typically a white to off-white crystalline solid.
- Melting Point: Around 150–160°C, depending on purity.
- Solubility: Soluble in common organic solvents like dichloromethane, tetrahydrofuran (THF), and dimethylformamide (DMF).
- Reactivity: The anhydride group is highly reactive toward nucleophiles, while the dihydroanthracene portion retains some aromatic stability.
Synthesis and Preparation
Common Synthetic Routes
The synthesis of 9,10-dihydroanthracene-9,10-α,β-succinic anhydride typically involves two main steps:
-
Hydrogenation of Anthracene:
Anthracene is partially hydrogenated using a catalyst like palladium on carbon (Pd/C) under controlled conditions. This selective hydrogenation targets the central benzene ring, forming 9,10-dihydroanthracene It's one of those things that adds up. Which is the point.. -
Anhydride Formation:
The dihydroanthracene intermediate is reacted with succinic anhydride or its precursor (succinic acid) in the presence of a dehydrating agent like acetic anhydride. This forms the cyclic anhydride bridge between the 9 and 10 positions.
Alternative Methods
Other synthetic approaches include:
- Diels-Alder Reaction: Using dihydroanthracene as a dienophile with a suitable diene to form the anhydride ring.
- Oxidative Coupling: Employing oxidizing agents to link the succinic acid groups to the anthracene core.
Applications and Uses
Organic Synthesis
This compound is primarily used as an intermediate in organic synthesis. Its dual reactivity—aromatic stability in the outer rings and the reactive anhydride—allows it to participate in a variety of reactions:
- Nucleophilic Acyl Substitution: The anhydride group reacts with amines, alcohols, or thiols to form amides, esters, or thioesters.
- Diels-Alder Reactions: The dihydroanthracene core can act as a dienophile in cycloaddition reactions.
- Polymer Precursors: Used in the synthesis of specialty polymers with tailored thermal or mechanical properties.
Pharmaceutical and Material Science
- Drug Development: The compound’s structure may serve as a scaffold for designing bioactive molecules.
- Functional Materials: Its rigid aromatic framework combined with the anhydride group makes it suitable for developing advanced materials, such as liquid crystals or organic semiconductors.
Safety and Handling
Hazards
- Toxicity: Limited data suggests moderate toxicity; avoid inhalation or skin contact.
- Reactivity: The anhydride group can cause irritation to eyes and respiratory systems.
- Flammability: Non-flammable under normal conditions but may decompose at high temperatures.
Storage and Precautions
- Store in a cool, dry place away from moisture and strong bases.
- Use personal protective equipment (PPE) such as gloves and goggles during
Storage and Precautions
Proper storage is critical to maintaining the stability and integrity of 9,10-dihydroanthracene-9,10-α,β-succinic anhydride. The compound should be kept in tightly sealed, amber-colored glass containers to protect it from light and moisture, which can hydrolyze the anhydride group. Storage areas must be cool (below 25°C), dry, and well-ventilated, with temperatures monitored to prevent thermal degradation. Compatibility is a key consideration: the compound should be isolated from strong acids, bases, and oxidizing agents, as these can trigger exothermic reactions or decomposition.
Handling requires strict adherence to safety protocols. Which means personnel should use chemical fume hoods to minimize inhalation risks and wear nitrile gloves, safety goggles, and lab coats to prevent skin or eye contact. Worth adding: spill containment kits should be readily available, and absorbent materials like vermiculite should be used to neutralize accidental releases. In case of skin exposure, immediate washing with soap and water is advised, followed by medical evaluation if irritation persists Small thing, real impact..
No fluff here — just what actually works.
Disposal must comply with local environmental regulations. Incineration in a licensed facility or chemical neutralization (e.g., hydrolysis under controlled conditions) are recommended methods. Waste streams should be tested for residual reactivity before disposal to avoid unintended environmental impacts But it adds up..
Conclusion
9,10-dihydroanthracene-9,10-α,β-succinic anhydride stands as a versatile and industrially significant compound, bridging the gap between organic synthesis and advanced material science. Its synthesis via hydrogenation and anhydride formation exemplifies the precision required in modern chemical manufacturing, while its dual reactivity—stemming from the aromatic core and the succinic anhydride—enables its use in pharmaceuticals, polymers, and functional materials. Still, its utility is accompanied by inherent hazards, necessitating rigorous safety measures during handling, storage, and disposal.
As research advances, this compound may tap into new applications in sustainable chemistry, such as biodegradable polymers or targeted drug delivery systems. By balancing innovation with caution, the chemical community can harness its potential responsibly, ensuring both scientific progress and occupational safety. Its role in Diels-Alder reactions and nucleophilic substitutions underscores its value in creating complex molecular architectures, while its thermal stability makes it a candidate for high-performance materials. In an era where interdisciplinary collaboration drives technological breakthroughs, 9,10-dihydroanthracene-9,10-α,β-succinic anhydride exemplifies the transformative power of carefully designed organic molecules That's the part that actually makes a difference..
Looking ahead, the future trajectory of 9,10-dihydroanthracene-9,10-α,β-succinic anhydride is intrinsically linked to advancements in chemical engineering and materials science. Computational modeling is poised to play a crucial role, enabling the prediction of its reactivity in novel Diels-Alder cycloadditions or as a monomer in complex polymer networks, potentially accelerating the discovery of tailored properties like enhanced thermal resistance or specific optical characteristics. What's more, the push towards sustainable chemistry will drive innovation in its synthesis. Exploring greener hydrogenation catalysts, solvent-free anhydride formation techniques, or even biocatalytic routes could significantly reduce the environmental footprint associated with its production, aligning with global sustainability goals Surprisingly effective..
Industrial scalability remains a key consideration. So while its synthesis is well-established, optimizing reaction conditions for higher yields, reduced energy consumption, and minimized waste generation is essential for broader adoption, particularly in high-volume applications like specialty polymers or advanced composite materials. Challenges such as catalyst longevity, efficient separation and purification of the anhydride product, and ensuring consistent batch-to-batch quality must be addressed through continuous process innovation and rigorous quality control protocols No workaround needed..
Not obvious, but once you see it — you'll see it everywhere The details matter here..
The compound's unique structure, combining a rigid anthracene-derived core with a reactive anhydride handle, continues to inspire research in supramolecular chemistry. Its potential to act as a building block for host-guest systems, molecular sensors, or components in organic electronics hinges on precise functionalization strategies that put to work both its aromatic stability and the reactivity of the anhydride ring. This dual nature, presenting both opportunities and challenges, underscores the need for interdisciplinary collaboration between synthetic organic chemists, materials scientists, and engineers to fully realize its potential.
All in all, 9,10-dihydroanthracene-9,10-α,β-succinic anhydride is far more than a synthetic intermediate; it is a foundational molecule with enduring relevance. Its journey from a carefully crafted organic compound to a component in up-to-date materials highlights the dynamic interplay between fundamental chemical principles and technological innovation. As research delves deeper into its reactivity, explores sustainable production pathways, and integrates it into sophisticated functional architectures, this compound is poised to remain a vital tool in the chemist's arsenal, driving progress across diverse scientific and industrial frontiers while demanding ongoing vigilance in its safe and responsible use. Its evolution mirrors the broader trajectory of chemistry itself: a discipline perpetually advancing through meticulous exploration, creative application, and a commitment to both discovery and safety Nothing fancy..