A Sample Of Brass Weighing 1.203 Grams
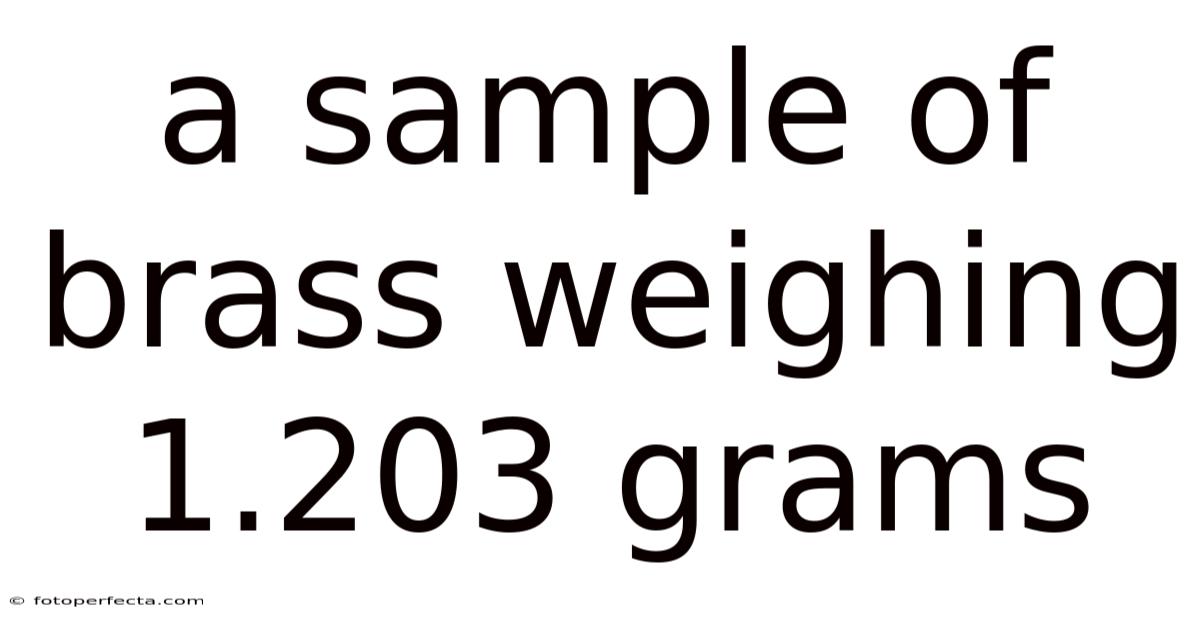
A Sample ofBrass Weighing 1.203 Grams: Precision in the Laboratory
In the meticulous world of scientific investigation and material analysis, even the smallest measurements hold profound significance. Consider a seemingly simple brass sample weighing exactly 1.203 grams. This precise figure, recorded with a high-precision analytical balance, represents far more than just a numerical value. It embodies the fundamental principles of measurement, the unique properties of this versatile alloy, and the critical role such data plays in research, quality control, and industrial applications. This article delves into the nature of this specific brass specimen, exploring its composition, the importance of its measured mass, and the scientific context surrounding its existence.
Brass, an alloy primarily composed of copper and zinc, has captivated human ingenuity for millennia. Its exact composition dictates its physical and mechanical properties, ranging from the soft, ductile yellow brasses to the harder, stronger alpha-beta and beta brasses. The specific ratio of copper to zinc determines characteristics like corrosion resistance, machinability, acoustic properties, and tensile strength. A brass sample weighing 1.203 grams is a tangible representation of this intricate balance. It could originate from a controlled laboratory synthesis, a precise manufacturing process, or perhaps a fragment retrieved from a historical artifact. Regardless of its origin, this specific mass is a crucial parameter, anchoring the sample within a defined context for further experimentation or analysis.
Measuring such a minute quantity requires sophisticated instrumentation. Analytical balances, capable of detecting changes as small as one milligram (0.001 grams), are the standard tool. Calibrating these balances meticulously and placing the sample within a draft-free environment ensures the accuracy of the 1.203 gram reading. This level of precision is non-negotiable. A discrepancy of even a fraction of a gram could lead to significant errors in subsequent calculations, such as determining density or performing stoichiometric ratios in chemical reactions. The recorded mass becomes the foundation upon which all other properties of the brass sample are built and verified.
Understanding the brass's density is a natural next step after establishing its mass. Density, defined as mass per unit volume (ρ = m/V), is a fundamental property that characterizes a material. For brass, typical densities range from approximately 8.4 to 8.7 grams per cubic centimeter (g/cm³), depending on the specific alloy composition and crystal structure. Using the measured mass of 1.203 grams, one can calculate the volume if the density is known. For instance, assuming a density of 8.5 g/cm³, the volume V would be calculated as:
V = m / ρ = 1.203 g / 8.5 g/cm³ ≈ 0.1415 cm³
This calculation transforms the abstract mass into a tangible spatial dimension, revealing how much space this small piece of brass occupies. This volume, combined with the mass, allows for further exploration of the sample's characteristics. It enables the determination of other properties like specific gravity (density relative to water) or provides essential data for processes like dissolution studies or thermal analysis.
The significance of this specific brass sample extends beyond its individual mass and density. Brass samples of defined mass and composition are indispensable tools in scientific research. They serve as calibration standards for analytical instruments, ensuring the reliability of measurements across different labs. In material science, they are used to study phase transformations, corrosion behavior under controlled conditions, or the impact of alloying elements. In quality control laboratories of manufacturing plants, samples of known mass and composition are tested against incoming raw materials or finished products to ensure they meet stringent specifications for strength, conductivity, or appearance. The 1.203 gram brass sample might be part of a batch tested for lead content, hardness, or acoustic resonance in musical instruments.
Moreover, the precise measurement of such a small mass highlights the evolution of weighing technology. From the delicate balances used by ancient alchemists to the ultra-precise electronic analytical balances of today, the ability to measure mass with such accuracy is a testament to human ingenuity. It allows scientists to work at scales invisible to the naked eye, pushing the boundaries of nanotechnology and materials engineering. The 1.203 gram brass sample is a microcosm of this precision, a small but vital piece in the vast puzzle of material characterization and scientific discovery.
Frequently Asked Questions (FAQ)
- Q: Why is the exact mass (1.203 g) so important?
- A: The exact mass is fundamental. It's used to calculate density, specific gravity, and other critical properties. It's also essential for stoichiometric calculations in chemical reactions involving the brass, and for comparing it to known standards or other samples.
- Q: How can such a small mass (1.203 g) be measured accurately?
- A: Using highly sensitive analytical balances, often with an accuracy of 0.1 mg (0.0001 g) or better. These balances are housed in controlled environments (like weighing rooms) to minimize air currents and vibrations.
- Q: What determines the density of brass?
- A: The density is primarily determined by the atomic masses of copper and zinc and their specific arrangement within the crystal lattice (phase structure). Different alloy compositions and processing methods (like cold working) can alter the density.
- Q: How is the volume of such a small brass sample calculated?
- A: By measuring its mass and knowing its density (ρ = m/V). Rearranging gives V = m/ρ. For example, with m = 1.203 g and ρ ≈
...8.5 g/cm³ (a typical value for brass), the calculated volume would be approximately 0.1415 cm³. This tiny volume, inferred from a precise mass, underscores how fundamental metrology—the science of measurement—is to quantifying the material world.
This principle scales up dramatically. In pharmaceutical research, milligram-to-microgram precision determines drug efficacy and safety. In environmental science, tracing parts-per-billion concentrations of pollutants relies on the same foundational accuracy. The 1.203-gram sample is not an isolated case but a representative instance of a universal practice: anchoring abstract theory to physical reality through exact numbers. It transforms a generic piece of metal into a defined entity with specific, testable properties, enabling reproducible experiments and valid comparisons across time and geography.
Ultimately, the story of this small brass sample is a story about standards, rigor, and trust. It reflects a shared commitment within the global scientific community to a common language of measurement. Every calibrated balance, every certified reference material, and every documented procedure exists to ensure that when a researcher in Tokyo measures a sample and a colleague in Berlin does the same, they are speaking the same quantitative dialect. This silent, rigorous framework is what allows cumulative progress to occur, building discovery upon discovery with confidence.
Conclusion
The humble 1.203-gram brass specimen thus embodies far more than its physical substance. It is a nexus where instrument calibration, material science, quality assurance, and the history of technological innovation converge. Its precise mass is the key that unlocks a cascade of derived properties and ensures its role as a reliable benchmark. In an era increasingly focused on the nano and micro scales, the ability to measure such small masses with unwavering accuracy remains a cornerstone of scientific integrity and industrial competence. It is a quiet testament to the fact that even the smallest number, when measured correctly, can hold the weight of immense knowledge and drive the engines of discovery forward.
Building on the foundation laid bythat calibrated brass mass, contemporary laboratories are re‑imagining how traceable accuracy can be achieved at the nanogram and even picogram levels. Micro‑electromechanical systems (MEMS) resonators, for instance, exploit the vibrational frequencies of microscopic cantilevers to infer mass changes with sub‑nanogram sensitivity, while quantum‑entangled photons are being harnessed in optomechanical setups that detect the recoil of individual atoms. These avant‑garde approaches inherit the same metrological rigor that once demanded a 1.203‑gram reference, but they translate it into a language of dynamic, real‑time sensing that can monitor chemical reactions as they unfold.
Parallel to technological innovation, there is a growing emphasis on sustainability within the metrology ecosystem. Manufacturers are now designing reference weights from recycled alloys and low‑impact manufacturing processes, reducing the carbon footprint associated with producing and certifying high‑purity standards. Moreover, the concept of “green calibration” encourages the use of portable, battery‑efficient instruments that can perform on‑site verifications, thereby minimizing the need for frequent transport of delicate artifacts to centralized calibration facilities.
The digital transformation of measurement science also reshapes how laboratories document and share their reference data. Cloud‑based metrology platforms aggregate calibration histories, environmental logs, and uncertainty budgets, enabling collaborative audits that transcend institutional borders. Machine‑learning algorithms can flag anomalous drift patterns before they compromise experimental integrity, offering a proactive safeguard that complements traditional statistical control charts.
Looking ahead, the integration of artificial intelligence with metrological workflows promises to refine uncertainty quantification to unprecedented levels. Predictive models trained on vast datasets of instrument behavior can suggest optimal calibration intervals, recommend cleaning protocols, and even forecast the performance of emerging sensor technologies under varying laboratory conditions. This convergence of analytics and measurement not only amplifies precision but also democratizes access to high‑quality standards, allowing smaller research groups to partake in the same calibrated certainty once reserved for elite national metrology institutes.
In sum, the legacy of the 1.203‑gram brass specimen continues to reverberate through every facet of modern measurement. From quantum‑enhanced resonators to eco‑conscious calibration practices, the quest for exactness fuels a cascade of innovation that propels science forward. By anchoring abstract concepts to tangible, reproducible standards, we preserve the trust that underpins collaborative discovery, ensuring that each new insight—no matter how minute—rests upon an unshakable foundation of verified accuracy.
Latest Posts
Latest Posts
-
6 10 6 Handling Multiple Exceptions Vending Machine Example
Mar 22, 2026
-
Post Test The Late Nineteenth Century Realism And Naturalism
Mar 22, 2026
-
Whose Corruption Did The Cartoons Of Thomas Nast Expose
Mar 22, 2026
-
Calculate Reconcile Your Checkbook Answer Key
Mar 22, 2026
-
Zig Ziglar 10 Rules For Success
Mar 22, 2026