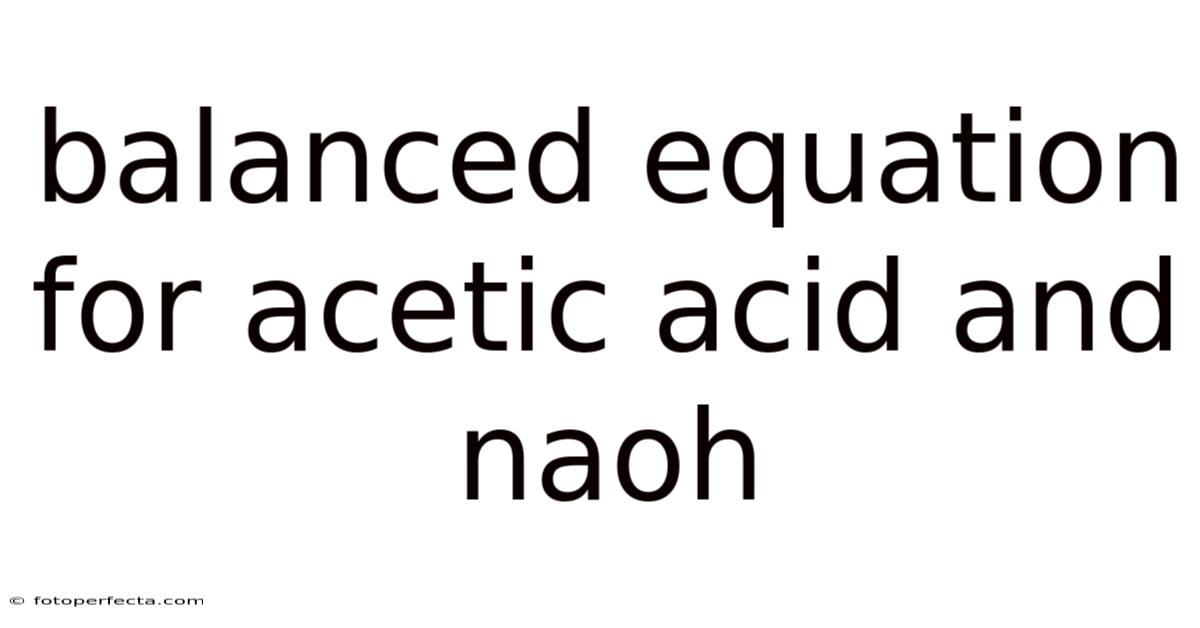The delicate interplay between acetic acid and sodium hydroxide underscores a fundamental principle within chemistry that has profound implications across various scientific disciplines. In real terms, these two substances, though seemingly distinct in their chemical natures, converge in a reaction that shapes the very foundation of many laboratory processes and natural phenomena. On top of that, acetic acid, a weak organic acid commonly found in vinegar, serves as a versatile component in numerous applications ranging from culinary uses to industrial formulations. Conversely, sodium hydroxide, a strong base widely utilized in cleaning agents and chemical synthesis, offers a contrasting role in altering pH dynamics. Their interaction exemplifies how seemingly opposing forces can harmonize to produce outcomes that are both practical and scientifically significant. In this context, understanding the precise mechanics of their combination becomes central, as it directly impacts outcomes in fields such as biochemistry, environmental science, and materials engineering. The task at hand is to dissect the balanced equation governing this reaction, unravel its components, and explore its ramifications thoroughly. This exploration not only clarifies the theoretical underpinnings but also highlights the practical applications that arise when these substances are employed together effectively. Here's the thing — such knowledge empowers individuals and professionals alike to deal with complex chemical systems with greater confidence, ensuring that the nuances of equilibrium, stoichiometry, and reaction kinetics are harnessed to achieve desired results. Through meticulous analysis, we uncover how foundational reactions serve as the building blocks upon which modern technological advancements are constructed, reinforcing the indispensable role of balanced equations in bridging theory and practice.
Balancing the chemical equation between acetic acid and sodium hydroxide demands precision, as even minor deviations can lead to imbalances with cascading consequences. The reaction in question, often termed the neutralization of acetic acid by sodium hydroxide, is a classic demonstration of acid-base chemistry. Acetic acid, with its formula CH3COOH, donates a proton (H+) to the hydroxide ion (OH-), facilitated by the hydroxide ion from sodium hydroxide (NaOH), resulting in acetate ion (CH3COO-) and water (H2O). Practically speaking, this process not only neutralizes the acidity of acetic acid but also transforms it into a salt of sodium acetate (CH3COONa), thereby restoring the pH balance of a solution previously dominated by acetic acid. On top of that, the balanced equation CH3COOH + NaOH → CH3COONa + H2O encapsulates this transformation succinctly yet powerfully, illustrating how stoichiometry governs the outcome. Each molecule of sodium hydroxide must react in a 1:1 molar ratio with acetic acid to ensure complete neutralization, a principle that underpins countless laboratory procedures. In practice, herein lies the critical challenge: ensuring that the quantities provided align perfectly to prevent incomplete reactions or excess reactants that could compromise the desired product. Which means such precision is not merely academic; it directly influences the efficiency and safety of chemical processes, from manufacturing pharmaceuticals to controlling environmental acidity levels. The equation serves as a blueprint, guiding practitioners in predicting outcomes and minimizing errors that could arise from miscalculations.
Subsequently, the concept of a balanced equation extends beyond mere numerical equality; it embodies the principle of conservation of mass and charge, ensuring that atoms and ions are accounted for accurately. In this case, the reactants must be consumed in exact proportions to yield the products without leftover reactants, which could lead to incomplete reactions or unexpected byproducts. This balance is particularly crucial in scenarios where precision is key, such as in pharmaceutical synthesis where even minor deviations might alter the efficacy of a drug. Beyond that, the equation’s structure offers insight into the reaction’s mechanism, revealing how the hydroxide ion attacks the protonated acetic acid molecule, disrupting its structure and facilitating the formation of the salt. Visualizing this process through the balanced equation allows for a clearer understanding of the intermediate steps, from proton abstraction to ion formation. Such clarity is invaluable for students and professionals who must interpret and apply chemical reactions in real-world contexts. The equation thus acts as both a guide and a verification tool, ensuring that theoretical knowledge translates effectively into practical application.
One might also consider the broader implications of balancing this equation within the context of larger chemical systems. Also, for instance, in industrial settings where acetic acid is utilized as a solvent or catalyst, maintaining stoichiometric accuracy ensures consistent performance. Conversely, in biological systems, where enzymes interact with acids and bases, the principles governing such reactions inform the design of therapeutic agents or ecological balance. The equation’s applicability extends beyond laboratory settings, influencing agricultural practices where pH adjustments are critical for plant health or water treatment processes.
On top of that, the ability to manipulate and interpret equations is essential for troubleshooting and optimizing chemical processes. Plus, engineers and scientists often rely on these principles to scale reactions, adjust conditions, or predict how changes in concentration affect yield. Day to day, this adaptability underscores why mastering the concept of balancing equations is a foundational skill across disciplines. It empowers learners to move from isolated experiments to comprehensive problem-solving, where precision directly correlates with successful outcomes.
In advanced studies, this concept also plays a vital role in exploring reaction kinetics and thermodynamics. By examining how variables such as temperature or catalyst presence shift the balance, researchers can refine methods to enhance efficiency and sustainability. The interplay between theory and application here highlights the dynamic nature of chemistry, where each equation serves as a stepping stone toward innovation That alone is useful..
At the end of the day, the significance of balancing equations extends far beyond the classroom—it is integral to advancing science, improving industrial processes, and shaping environmental stewardship. By consistently applying this principle, we not only deepen our understanding of chemical behavior but also contribute to solutions that benefit society at large.
Conclusion: Balancing equations is more than a technical exercise; it is a cornerstone of scientific inquiry and practical application, reinforcing the value of precision in both learning and real-world endeavors.
This foundational competence also cultivates a particular mode of systematic thinking—one that values conservation, proportionality, and logical sequence. Which means such a mindset transcends chemistry, proving invaluable in fields like materials science, environmental policy, and even computational modeling, where systems must be quantified and balanced. Beyond that, in an era of interdisciplinary collaboration, the ability to precisely communicate quantitative relationships through a universal symbolic language like chemical equations breaks down barriers between specialties, enabling cohesive progress on multifaceted challenges Worth knowing..
The bottom line: the disciplined practice of balancing equations instills more than procedural accuracy; it nurtures intellectual rigor and a commitment to empirical truth. It reminds us that even the most complex phenomena can be understood through careful, balanced analysis—a principle as relevant to decoding a metabolic pathway as it is to designing a circular chemical economy. By mastering this essential skill, we equip ourselves not only to interpret the existing world but also to innovate responsibly within it.
Conclusion: Which means, the act of balancing chemical equations is a profound exercise in intellectual integrity and practical foresight. It is the silent, steady grammar upon which the language of chemistry is built, empowering us to decode nature’s processes and author sustainable solutions. In every balanced equation lies a microcosm of order—a testament to the fact that precision in representation is the first and most crucial step toward mastery in both science and its responsible application.
Conclusion: That's why, the act of balancing chemical equations is a profound exercise in intellectual integrity and practical foresight. It is the silent, steady grammar upon which the language of chemistry is built, empowering us to decode nature’s processes and author sustainable solutions. In every balanced equation lies a microcosm of order—a testament to the fact that precision in representation is the first and most crucial step toward mastery in both science and its responsible application Which is the point..
Beyond its direct utility in stoichiometric calculations, the skill of balancing equations fosters critical thinking applicable to a wide range of problems. On top of that, it encourages a structured approach to problem-solving, demanding careful consideration of inputs and outputs, and promoting a systematic approach to identifying and resolving imbalances. This analytical mindset is directly transferable to fields requiring quantitative reasoning, from economics and engineering to biology and medicine. The ability to dissect a complex system into its constituent parts, quantify their relationships, and determine the necessary adjustments is a fundamental skill for navigating an increasingly data-driven world.
Worth adding, the continuous refinement of balancing techniques, driven by advancements in computational chemistry and analytical instrumentation, underscores the dynamic and evolving nature of scientific knowledge. Modern software tools automate many of the tedious calculations, but a deep understanding of the underlying principles remains essential for interpreting results, troubleshooting errors, and designing new experiments. The ability to critically evaluate these tools and apply them effectively is a crucial skill for any aspiring chemist or scientist Most people skip this — try not to..
Finally, the emphasis on conservation principles inherent in balancing equations resonates deeply with contemporary concerns about sustainability. Practically speaking, the chemical industry is increasingly focused on minimizing waste, maximizing resource utilization, and developing environmentally benign processes. It allows us to predict product yields, minimize byproducts, and ultimately contribute to a circular economy where resources are valued and waste is minimized. On the flip side, a firm grasp of stoichiometry and reaction equilibrium, built upon the foundation of balanced equations, is indispensable for designing and optimizing these sustainable chemical pathways. The seemingly simple act of balancing an equation, therefore, becomes a powerful symbol of our commitment to responsible innovation and a healthier planet.