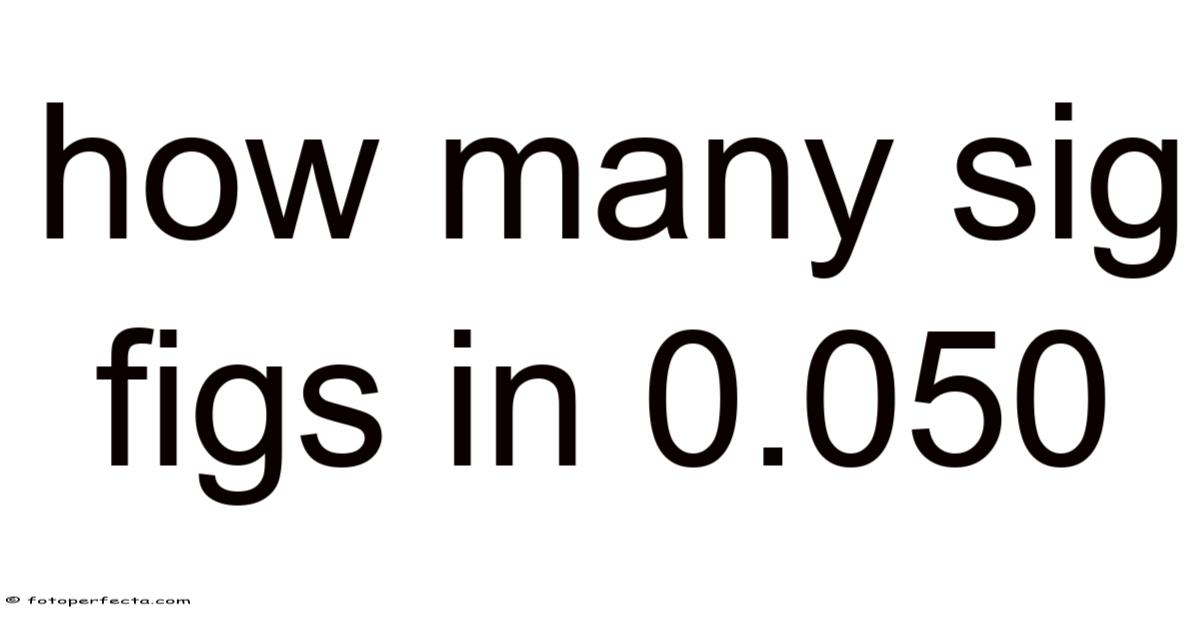How Many Sig Figs in 0.050?
Significant figures, often abbreviated as "sig figs," represent the meaningful digits in a number that contribute to its precision. But when working with measurements in scientific contexts, understanding significant figures is crucial for maintaining accuracy and communicating the precision of your results. The number 0.050 presents an interesting case for determining significant figures, as it contains both leading and trailing zeros that require careful analysis according to established rules Practical, not theoretical..
What Are Significant Figures?
Significant figures are the digits in a number that carry meaning contributing to its measurement precision. This includes all digits except:
- Leading zeros (zeros before non-zero digits)
- Placeholder zeros (zeros that are only placeholders for positioning)
In scientific calculations, significant figures help communicate the precision of measurements and see to it that calculations don't imply greater accuracy than what was actually measured. The number of significant figures in a measurement indicates the precision of the measuring instrument used.
Rules for Determining Significant Figures
To determine how many significant figures are in a number like 0.050, we need to understand the established rules:
-
Non-zero digits are always significant. To give you an idea, in 123, all three digits are significant Not complicated — just consistent. Which is the point..
-
Any zeros between significant digits are significant. Here's one way to look at it: in 101, all three digits are significant Not complicated — just consistent. Took long enough..
-
Leading zeros are never significant. They merely indicate the position of the decimal point. As an example, in 0.0012, only the 1 and 2 are significant.
-
Trailing zeros are significant only if the number contains a decimal point. Here's one way to look at it: in 1200, the trailing zeros may or may not be significant (ambiguous), but in 1200., they are significant But it adds up..
-
In numbers less than one, all zeros after the decimal point and before a non-zero digit are not significant. Take this: in 0.0045, only the 4 and 5 are significant.
-
For numbers with trailing zeros after a decimal point, all zeros are significant. As an example, in 4.500, all four digits are significant But it adds up..
Analyzing 0.050 Step by Step
Let's apply these rules to determine how many significant figures are in 0.050:
-
First, identify the non-zero digits: In 0.050, we have the digit 5 Most people skip this — try not to. Simple as that..
-
Examine the zeros:
- The first zero after the decimal point (0.050) is a leading zero and not significant.
- The zero between the decimal point and the 5 (0.050) is also a leading zero and not significant.
- The zero after the 5 (0.050) is a trailing zero after a decimal point, making it significant.
-
Which means, the significant digits in 0.050 are:
- The digit 5 (significant)
- The final zero (significant because it's a trailing zero after a decimal point)
So, 0.050 has two significant figures.
Common Mistakes and Misconceptions
When determining significant figures in numbers like 0.050, several common mistakes often occur:
-
Counting all zeros as significant: Some people mistakenly count all zeros in a number as significant, which would incorrectly lead them to believe 0.050 has three significant figures.
-
Ignoring the significance of trailing zeros after a decimal: The trailing zero in 0.050 is significant because it indicates precision to the thousandths place. Without this zero, the number would be 0.05, which has only one significant figure and implies less precision.
-
Confusing decimal points with significant figures: The presence of a decimal point is crucial in determining whether trailing zeros are significant. In 0.050, the decimal point makes the final zero significant, whereas in 50, the zero might not be significant Worth keeping that in mind..
-
Misapplying rules for scientific notation: When converting 0.050 to scientific notation (5.0 × 10^-2), it becomes clearer that there are two significant figures.
Scientific Notation and Significant Figures
Scientific notation can often make the number of significant figures more obvious. Let's express 0.050 in scientific notation:
0.050 = 5.0 × 10^-2
In scientific notation, all digits in the coefficient (5.In practice, 0 in this case) are significant. Even so, the exponent (10^-2) simply indicates the position of the decimal point and does not affect the significant figures. This clearly shows that 0.050 has two significant figures Easy to understand, harder to ignore..
Scientific notation is particularly useful for numbers with many leading or trailing zeros, as it eliminates ambiguity about which digits are significant It's one of those things that adds up..
Practical Applications of Significant Figures
Understanding significant figures has important practical applications across various scientific and technical fields:
-
Chemistry: In chemical reactions, the precision of measurements affects the accuracy of results. To give you an idea, if you're measuring 0.050 grams of a substance, this indicates a precision to the thousandths place, which might be crucial for stoichiometric calculations Less friction, more output..
-
Physics: When reporting experimental results, significant figures communicate the precision of measurements. A measurement of 0.050 meters (5.0 cm) implies greater precision than 0.05 meters (5 cm).
-
Engineering: In engineering calculations, significant figures help see to it that designs and specifications maintain appropriate precision. Using 0.050 rather than 0.05 could represent a critical difference in tolerances.
-
Medicine: Dosage calculations often require careful attention to significant figures to ensure patient safety. A dosage of 0.050 mg has different implications than 0.05 mg No workaround needed..
-
Finance: While financial calculations may use different conventions, understanding significant figures helps
ensure consistency and accuracy in reporting values. 5% versus 12.Worth adding: for example, reporting a profit margin as 12. 50% conveys different levels of precision and should be considered carefully Small thing, real impact..
Common Pitfalls to Avoid
Despite the relatively straightforward rules, significant figures can still be a source of confusion. Here are some common mistakes to watch out for:
-
Rounding errors: Incorrectly rounding numbers during calculations can lead to an inaccurate number of significant figures in the final result. Always carry extra digits during intermediate calculations and round only the final answer to the appropriate number of significant figures.
-
Addition and subtraction: When adding or subtracting numbers, the result should be rounded to the same number of decimal places as the number with the fewest decimal places. Significant figures are also adjusted accordingly.
-
Multiplication and division: In multiplication and division, the result should have the same number of significant figures as the number with the fewest significant figures in the original calculation Still holds up..
-
Significant figures in addition/subtraction: When adding or subtracting numbers, the result should be rounded to the least number of significant figures present in the numbers being added or subtracted.
Conclusion
Mastering the concept of significant figures is fundamental to accurate scientific and technical communication. It's not simply about memorizing rules, but about understanding the limitations of measurement and the importance of conveying precision effectively. So remember, significant figures are not arbitrary; they are a vital tool for communicating the reliability and precision of your data and calculations – a skill essential for success in any field that relies on quantitative analysis. By diligently applying the rules and being aware of common pitfalls, you can make sure your work is presented with the appropriate level of accuracy and clarity. Paying attention to these details demonstrates rigor and professionalism, ultimately leading to more trustworthy and dependable results.