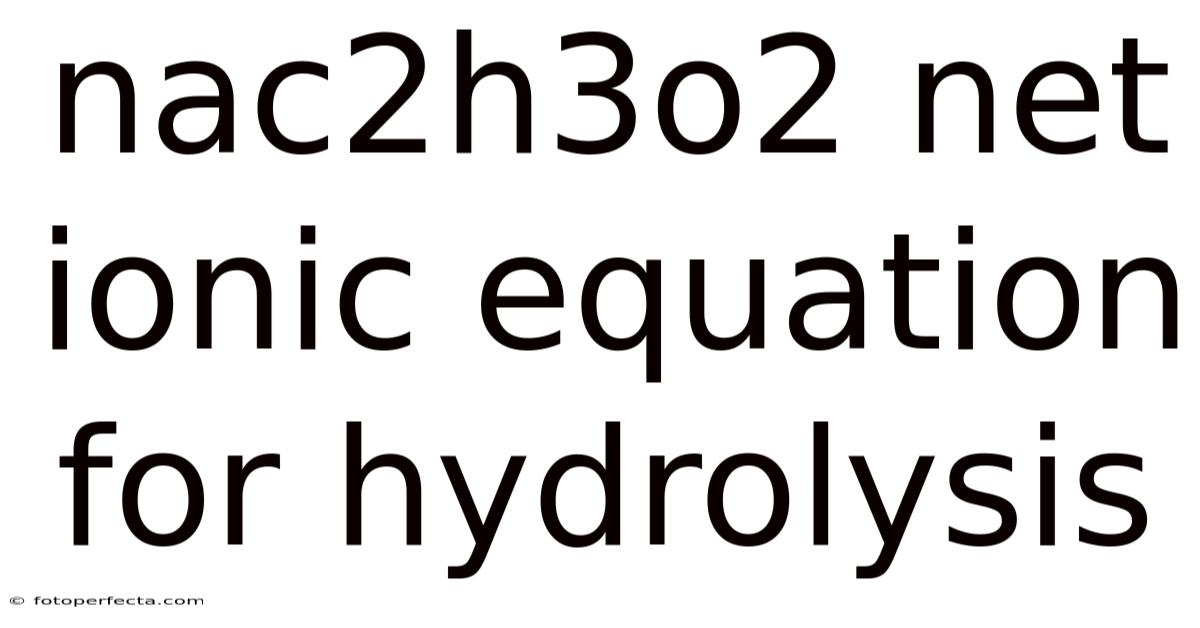The nac2h3o2 net ionic equation for hydrolysis reveals how sodium acetate interacts with water to form a mildly basic solution, a fundamental concept in acid-base chemistry that bridges theoretical equilibrium principles with real-world laboratory behavior. When sodium acetate dissolves in water, it undergoes a hydrolysis reaction where only the acetate ion participates in proton transfer, leaving sodium ions as inactive spectators. Understanding this process not only clarifies why salt solutions can alter pH but also strengthens your grasp of conjugate acid-base pairs, equilibrium shifts, and net ionic notation. By breaking down each step and examining the underlying chemical principles, you will gain a clear, practical understanding of how weak acid salts behave in aqueous environments The details matter here. No workaround needed..
Introduction
Sodium acetate, commonly written as NaC₂H₃O₂ or NaCH₃COO, is an ionic compound formed from the neutralization of acetic acid (a weak acid) and sodium hydroxide (a strong base). In solid form, it exists as a crystalline lattice of sodium cations and acetate anions. Once introduced to water, the compound dissociates completely into its constituent ions. Even so, dissociation alone does not explain the chemical behavior of the resulting solution. The true chemical activity emerges during hydrolysis, a process where dissolved ions react with water molecules to shift the pH away from neutrality. For sodium acetate, this means the acetate ion accepts a proton from water, generating hydroxide ions and making the solution slightly alkaline. Grasping this transformation requires distinguishing between full dissociation, spectator ions, and the actual reactive species that appear in the nac2h3o2 net ionic equation for hydrolysis. Mastering this distinction is essential for success in general chemistry, analytical methods, and buffer preparation Practical, not theoretical..
Steps
Writing a net ionic equation requires filtering out non-participating species and focusing only on the chemical change. Follow these systematic steps to derive the correct expression:
- Write the complete dissociation equation: When solid sodium acetate dissolves, it separates into aqueous ions: NaC₂H₃O₂(s) → Na⁺(aq) + C₂H₃O₂⁻(aq)
- Identify the hydrolysis reaction: The acetate ion acts as a weak base and reacts reversibly with water: C₂H₃O₂⁻(aq) + H₂O(l) ⇌ HC₂H₃O₂(aq) + OH⁻(aq)
- Determine spectator ions: Sodium ions (Na⁺) originate from a strong base and do not react with water. They remain unchanged throughout the process, qualifying them as spectators.
- Remove spectators to form the net ionic equation: Eliminating Na⁺ leaves only the reacting species: C₂H₃O₂⁻(aq) + H₂O(l) ⇌ HC₂H₃O₂(aq) + OH⁻(aq)
- Verify charge and mass balance: Both sides contain one acetate-derived group, two hydrogen atoms from water, and a net charge of −1. The equation is balanced and chemically accurate.
This streamlined approach ensures you capture only the essential proton-transfer event, which is the core of the hydrolysis process. Practicing this method with other salts will quickly build your confidence in predicting aqueous behavior Surprisingly effective..
Scientific Explanation
The hydrolysis of acetate ions is governed by Brønsted-Lowry acid-base theory and the concept of conjugate pairs. Acetic acid (HC₂H₃O₂) is classified as a weak acid because it only partially ionizes in water. So naturally, its conjugate base, the acetate ion, retains a measurable tendency to accept protons. When placed in water, the acetate ion competes with hydroxide ions for available protons, pulling the autoionization equilibrium of water to the right. This shift produces molecular acetic acid and free hydroxide ions, which accumulate in solution.
The extent of this reaction is quantified by the base dissociation constant, K_b. Since acetate is the conjugate base of acetic acid, its K_b can be calculated using the ion-product constant of water (K_w = 1.0 × 10⁻¹⁴ at 25°C) and the acid dissociation constant of acetic acid (K_a ≈ 1.8 × 10⁻⁵): K_b = K_w / K_a ≈ 5 Took long enough..
Worth pausing on this one.
Although this value is small, it is large enough to generate a detectable concentration of OH⁻ ions. The double arrow (⇌) in the equation emphasizes that the reaction reaches a dynamic equilibrium rather than proceeding to completion. But temperature, concentration, and the presence of other ions can shift this equilibrium according to Le Chatelier’s principle, but under standard laboratory conditions, the hydrolysis remains partial and predictable. Recognizing that weak acid salts always produce basic solutions while weak base salts produce acidic solutions provides a reliable mental shortcut for exam questions and experimental planning.
Why the Solution Becomes Basic
The production of hydroxide ions directly explains why sodium acetate solutions register a pH above 7. In pure water, the concentration of H⁺ and OH⁻ ions remains equal at 1.0 × 10⁻⁷ M, yielding a neutral pH. When acetate ions hydrolyze, they consume a small fraction of water molecules and release OH⁻ ions, disrupting this balance. As hydroxide concentration rises, the solution becomes alkaline And it works..
Several practical factors influence the final pH:
- Initial concentration: Higher sodium acetate concentrations yield more acetate ions, increasing OH⁻ production and raising pH. So - Temperature: Since K_w increases with temperature, the baseline neutrality point shifts, and hydrolysis rates may change slightly. Here's the thing — - Common ion effect: Adding acetic acid or another acetate salt suppresses hydrolysis by shifting equilibrium leftward, demonstrating how buffer systems stabilize pH. - Ionic strength: High concentrations of unrelated electrolytes can slightly alter activity coefficients, though this effect is typically negligible in introductory contexts.
Understanding this behavior is essential in fields ranging from analytical chemistry to food preservation, where sodium acetate is used to control acidity, stabilize reactions, or create mild alkaline environments without introducing harsh bases. The predictable nature of the nac2h3o2 net ionic equation for hydrolysis makes it a reliable tool for designing experiments that require precise pH control Nothing fancy..
Frequently Asked Questions
Q: Why doesn’t the sodium ion participate in hydrolysis?
Sodium ions are the conjugate acid of sodium hydroxide, a strong base. Strong bases dissociate completely and their conjugate acids are so weak that they show no tendency to accept hydroxide or donate protons in water. So, Na⁺ remains chemically inert and is excluded from the net ionic equation.
Q: Can I write the equation using CH₃COO⁻ instead of C₂H₃O₂⁻?
Yes. Both notations represent the acetate ion. The formula CH₃COO⁻ emphasizes the molecular structure, while C₂H₃O₂⁻ is the condensed empirical form. Both are chemically identical and acceptable in academic and laboratory settings.
Q: Does this hydrolysis reaction go to completion?
No. The reaction is reversible and reaches equilibrium quickly. The small K_b value indicates that only a tiny fraction of acetate ions convert to acetic acid and hydroxide at any given moment.
Q: How is this different from the hydrolysis of ammonium chloride?
Ammonium chloride produces an acidic solution because the ammonium ion (NH₄⁺) donates a proton to water, generating H₃O⁺. Sodium acetate does the opposite: the acetate ion accepts a proton, generating OH⁻. Both are hydrolysis reactions, but they move pH in opposite directions due to the nature of the parent acid or base The details matter here..
Conclusion
Mastering the nac2h3o2 net ionic equation for hydrolysis equips you with a clear framework for predicting how salt solutions interact with water. By recognizing that only the acetate ion actively participates in proton transfer while sodium ions remain spectators,
you can confidently predict the resulting alkaline shift and calculate equilibrium concentrations with precision. Whether you are troubleshooting a titration curve, designing a pH-stable formulation, or interpreting laboratory data, the hydrolysis behavior of sodium acetate serves as a textbook example of salt-water interactions. At the end of the day, mastering this reaction reinforces a core principle of aqueous chemistry: the properties of a dissolved salt are dictated not by the compound itself, but by the relative strengths of its parent acid and base. This foundational understanding bridges theoretical acid-base chemistry with real-world applications, from buffer formulation to industrial process control. With this insight, you are well-equipped to deal with more complex equilibria and apply acid-base theory with confidence in both academic and professional settings.