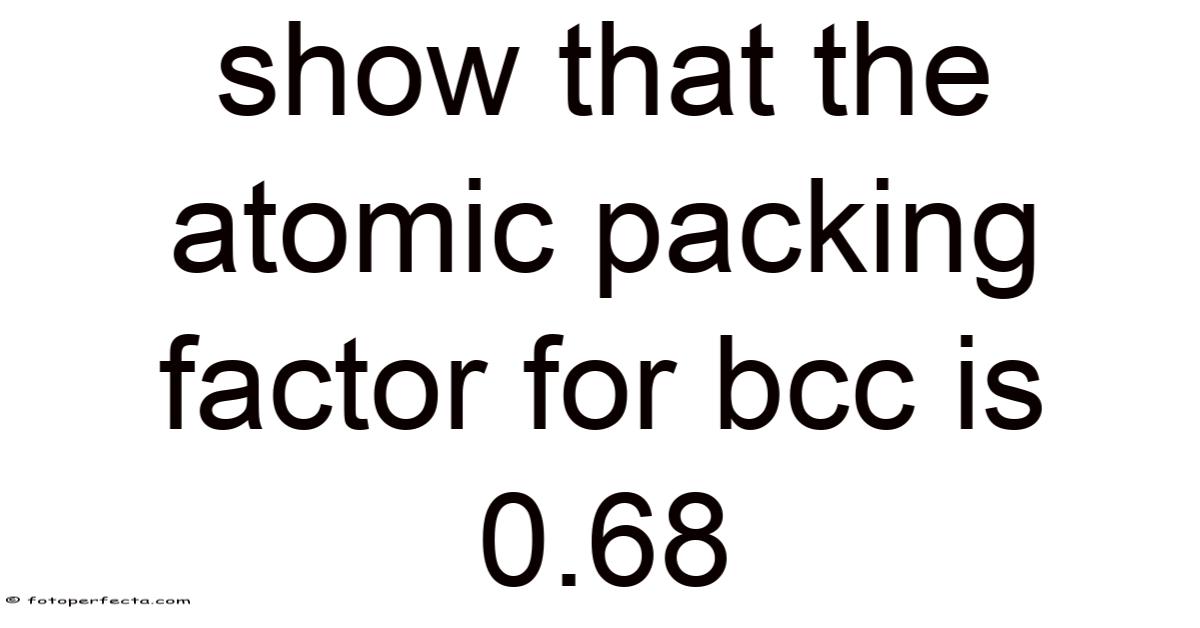The Atomic Packing Factor for Body-Centered Cubic (BCC) Structures: Demonstrating the 0.68 Value
The atomic packing factor (APF) is a fundamental concept in materials science that quantifies how efficiently atoms are arranged within a crystal structure. Which means for body-centered cubic (BCC) structures, this value is precisely 0. Even so, 68, which has significant implications for material properties and behavior. Understanding how this value is derived provides insight into the relationship between atomic arrangement and material characteristics.
Understanding Atomic Packing Factor
The atomic packing factor represents the fraction of volume in a crystal structure that is occupied by atoms. It is calculated using the formula:
APF = (Volume of atoms in the unit cell) / (Total volume of the unit cell)
This dimensionless value ranges from 0 to 1, with higher values indicating more efficient packing of atoms. Different crystal structures exhibit different atomic packing factors, which directly influence material properties such as density, mechanical strength, thermal conductivity, and diffusion rates.
Body-Centered Cubic Structure
The body-centered cubic (BCC) structure is one of the most common crystal structures found in metals. In a BCC structure:
- There is one atom at each corner of the cube
- There is one atom at the center of the cube
- Each corner atom is shared by 8 adjacent unit cells
- The center atom is entirely contained within the unit cell
To calculate the number of atoms per unit
###Calculating the Atomic Packing Factor for BCC
To calculate the number of atoms per unit cell in a BCC structure:
- Each of the 8 corner atoms contributes 1/8 of its volume to the unit cell (since it is shared among 8 adjacent cells).
- The center atom is entirely within the unit cell.
- Total atoms per unit cell = (8 \times \frac{1}{8} + 1 = 2).
Next, the volume occupied by these atoms is determined. Assuming each atom is a sphere with radius (r), the total volume of atoms in the unit cell is:
[
\text{Volume of atoms} = 2 \times \left(\frac{4}{3}\pi r^3\right) = \frac{8}{3}\pi r^3.
]
The unit cell’s edge length ((a)) must be related to the atomic radius. In a BCC structure, atoms touch along the body
This relationship also clarifies why BCC metals typically exhibit a lower density than FCC or HCP metals, despite having similar atomic radii. Because of that, because only 68 % of the available space is filled, the overall mass per unit volume is reduced, which can be advantageous when weight savings are critical. Beyond that, the relatively open lattice provides more free volume for dislocation motion, especially at elevated temperatures, contributing to the characteristic ductility of BCC alloys such as iron and molybdenum.
In practical terms, designers exploit the 0.68 APF when selecting materials for components that must balance strength with formability, such as high‑strength steel used in automotive frames or titanium‑based BCC alloys in aerospace structures. The known packing efficiency also informs calculations of theoretical density, which are essential for predicting material behavior in simulations and for optimizing manufacturing processes like powder metallurgy or additive manufacturing, where controlling atomic packing can influence final part properties Simple as that..
So, to summarize, the atomic packing factor of 0.68 for body‑centered cubic structures quantifies how efficiently atoms occupy space within the unit cell, directly linking microscopic arrangement to macroscopic
properties. This relationship also clarifies why BCC metals typically exhibit a lower density than FCC or HCP metals, despite having similar atomic radii. Because only 68 % of the available space is filled, the overall mass per unit volume is reduced, which can be advantageous when weight savings are critical. On top of that, the relatively open lattice provides more free volume for dislocation motion, especially at elevated temperatures, contributing to the characteristic ductility of BCC alloys such as iron and molybdenum Small thing, real impact. That alone is useful..
In practical terms, designers exploit the 0.Consider this: 68 APF when selecting materials for components that must balance strength with formability, such as high‑strength steel used in automotive frames or titanium‑based BCC alloys in aerospace structures. The known packing efficiency also informs calculations of theoretical density, which are essential for predicting material behavior in simulations and for optimizing manufacturing processes like powder metallurgy or additive manufacturing, where controlling atomic packing can influence final part properties But it adds up..
All in all, the atomic packing factor of 0.Even so, understanding this relationship empowers engineers and materials scientists to tailor alloy properties for specific applications, from lightweight aerospace components to solid structural steels. That's why 68 for body‑centered cubic structures quantifies how efficiently atoms occupy space within the unit cell, directly linking microscopic arrangement to macroscopic performance. As research advances, further insights into BCC behavior—especially under extreme conditions or in nanostructured forms—will continue to refine our ability to design materials with precision, reinforcing the central role of crystal structure in modern materials engineering.
The interplay between structure and material properties remains central to advancing engineering solutions Most people skip this — try not to..
Boiling it down, precise material selection hinges on such fundamental insights, bridging microscale precision with macro-scale application.
Thus, mastering these concepts ensures alignment with evolving technological demands.
The influence of the 0.68 APF extends beyond static properties; it also governs dynamic phenomena such as diffusion and phase stability. Because the BCC lattice offers more interstitial sites than the close‑packed FCC or HCP arrangements, atoms can migrate more readily, which underpins the high-temperature creep resistance of many BCC alloys. This behavior is exploited in high‑temperature structural steels and refractory metals, where maintaining dimensional stability under prolonged thermal exposure is critical.
From a thermodynamic perspective, the relatively open BCC framework lowers the cohesive energy density compared to FCC metals, which in turn affects melting points and phase transition temperatures. So naturally, designers often pair BCC metals with alloying elements that either reinforce the lattice (e.Which means g. , adding interstitial carbon to steel) or create solid solutions that modify the APF subtly, thereby tuning mechanical properties without compromising manufacturability And it works..
In additive manufacturing, the rapid solidification cycles can trap non‑equilibrium BCC phases, leading to unique microstructures that exhibit superior strength-to-weight ratios. By carefully controlling the cooling rate and post‑processing heat treatment, engineers can manipulate the fraction of BCC versus FCC phases, leveraging the 0.68 APF as a quantitative guide to predict density changes, residual stresses, and final part performance.
The bottom line: the atomic packing factor is more than a static number; it is a compass that directs the entire materials lifecycle—from alloy design and process selection to performance prediction and failure analysis. By integrating APF considerations with modern computational tools, such as density functional theory and machine‑learning models, the field is moving toward an era where materials can be engineered at the atomic level for targeted applications.
Conclusion
The body‑centered cubic structure, characterized by an atomic packing factor of 0.68, exemplifies how subtle differences in atomic arrangement translate into pronounced variations in density, strength, ductility, and thermal behavior. Recognizing and exploiting this relationship enables the rational design of lightweight, high‑performance components across aerospace, automotive, and energy sectors. As additive manufacturing and nanostructuring techniques mature, the ability to control and predict BCC packing will become even more key, ensuring that materials continue to meet the escalating demands of modern engineering.
The influence of the atomic packing factor within the BCC lattice extends far beyond simple structural description—it shapes the very performance and adaptability of materials in demanding environments. The interplay between lattice geometry and material properties underscores the importance of precision in both formulation and processing. And as research progresses, the integration of advanced modeling techniques promises to refine our grasp of these relationships, opening new avenues for material innovation. Now, by understanding how this factor governs diffusion, phase stability, and mechanical behavior, engineers can strategically tailor alloys for specific applications. Also, the journey from theoretical insight to practical implementation highlights a clear trajectory: a deeper appreciation of BCC packing will drive the next generation of high-performance materials. In this evolving landscape, the atomic-level manipulation of packing factors remains a cornerstone for advancing technology across industries.