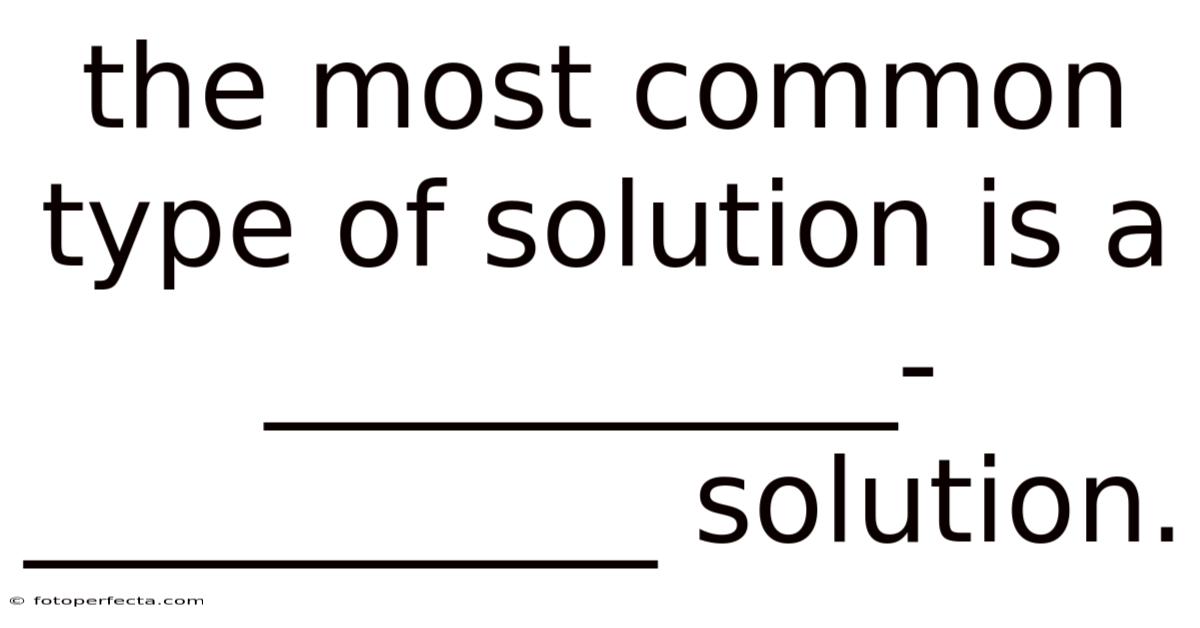Understanding the most common type of solution is essential for anyone looking to grasp the fundamentals of chemistry and its applications. In this article, we will delve into the significance of a solvent and explore why it plays a pivotal role in various scientific processes. By the end of this discussion, you will have a clear understanding of how this solution type influences reactions and everyday life.
The concept of a solution is foundational in the study of chemistry. A solution is essentially a homogeneous mixture where one substance is dissolved in another. The most common type of solution is a aqueous solution, which is composed primarily of water. This type of solution is not only prevalent in nature but also widely used in laboratories and industrial applications. The importance of understanding this solution type lies in its versatility and the numerous roles it plays in both scientific research and practical scenarios.
When we talk about solutions, we often refer to the properties that define them. One of the key characteristics of an aqueous solution is its ability to conduct electricity when a salt or electrolyte is dissolved. This property makes it incredibly useful in various fields, including medicine, food science, and environmental studies. For instance, in the pharmaceutical industry, aqueous solutions are essential for the formulation of medications. They help ensure that drugs are delivered effectively to the body, enhancing their therapeutic effects.
In the realm of education, it is crucial to highlight the role of solutions in experiments. Students often learn about chemical reactions through the manipulation of solutions. By understanding how different substances interact in these mixtures, learners can predict outcomes and gain deeper insights into the principles of chemistry. This hands-on approach not only reinforces theoretical knowledge but also builds confidence in applying concepts in real-world situations.
Moreover, the importance of solutions extends beyond the classroom. In agriculture, for example, water solutions are vital for crop growth. Farmers rely on these solutions to provide essential nutrients to plants, ensuring healthy development and bountiful harvests. Similarly, in the food industry, solution-based processes are used to create beverages, sauces, and desserts that enhance taste and texture. These applications underscore the significance of understanding aqueous solutions in everyday life.
To further clarify the concept, let’s break down the key components of a solvent and its role in forming solutions. A solvent is the substance that dissolves another substance, known as the solute. The choice of solvent is critical, as it affects the properties of the resulting solution. Common solvents include water, ethanol, and alcohols. When a solute is introduced into a solvent, it begins to disperse evenly throughout the liquid, creating a uniform mixture. This process is fundamental to the formation of aqueous solutions, which are prevalent in numerous scientific and practical applications.
Understanding the structure of solutions can also provide valuable insights. In a aqueous solution, the ions and molecules of the solute interact with the water molecules, leading to a dynamic equilibrium. This interaction is what gives solutions their unique characteristics, such as solubility and conductivity. For example, when you mix sugar in water, the sugar molecules dissolve due to their interaction with the water molecules. This process is not only fascinating but also essential for the functioning of many biological systems.
In addition to their scientific importance, solutions also play a significant role in environmental science. Water is the primary solvent in our planet, and its ability to dissolve a wide range of substances makes it indispensable for sustaining life. From pollution control to water purification, aqueous solutions are at the forefront of efforts to protect our environment. By recognizing the role of solutions in these processes, we can appreciate their broader implications beyond the laboratory.
As we explore the world of solutions, it becomes clear that their impact is vast and varied. Whether in the formulation of medicines, the production of food, or the management of natural resources, aqueous solutions are integral to our daily lives. This understanding not only enhances our knowledge but also empowers us to make informed decisions in various aspects of life.
In conclusion, the most common type of solution is an aqueous solution. This type of mixture plays a vital role in numerous scientific and practical applications. By recognizing the importance of solvents and the interactions between solutes and solvents, we can better appreciate the complexities of chemistry. As we continue to learn and apply this knowledge, we open the door to new possibilities and innovations. Embrace the journey of understanding, and let the power of solutions guide your curiosity and creativity.
Beyond these fundamentals, the behavior of solutions becomes even more intriguing when we consider factors like concentration, temperature, and pressure. These variables dictate not only how much solute can dissolve—the solubility limit—but also the rate of dissolution and the potential for forming supersaturated states. Such principles are harnessed in industrial crystallization, where precise control yields pure compounds, and in pharmacology, where the bioavailability of a drug hinges on its solubility in bodily fluids. The concept of ideal versus non-ideal solutions further reveals the nuanced intermolecular forces at play, explaining phenomena like boiling point elevation and freezing point depression that are critical for applications ranging from antifreeze formulation to food preservation.
The microscopic interactions within a solution also lay the groundwork for understanding colligative properties and electrochemical systems. In electrolyte solutions, the complete dissociation of ions enables electrical conductivity, a property exploited in batteries, electroplating, and even neural signaling. Conversely, the careful manipulation of non-electrolyte solutions allows for the creation of colloids and suspensions, which have distinct properties vital for products like paints, cosmetics, and diagnostic reagents. This spectrum—from true solutions to heterogeneous dispersions—demonstrates the remarkable versatility of dissolved systems across material science.
Looking ahead, the principles of solution chemistry are pivotal in addressing global challenges. The development of greener solvents, such as supercritical carbon dioxide or ionic liquids, aims to replace volatile organic compounds in industrial processes. In energy research, novel electrolyte solutions are being engineered for next-generation batteries and fuel cells. Furthermore, environmental remediation strategies often rely on tailored aqueous solutions to extract heavy metals or degrade persistent pollutants. Thus, the humble mixture of solute and solvent is not merely a laboratory curiosity but a dynamic platform for innovation.
In conclusion, while the aqueous solution is the most familiar, the study of solutions encompasses a vast landscape of interactions and applications that shape technology, medicine, and environmental stewardship. By appreciating both the simple elegance of dissolution and the complex science of molecular interactions, we gain tools to solve pressing problems and create sustainable technologies. The journey into the world of solutions is, ultimately, a journey into the very mechanisms of change and transformation that define our material world.
The future of solution chemistry promises even more profound advancements. Computational modeling is rapidly accelerating our ability to predict and manipulate solution behavior, allowing for the design of materials with tailored properties. This extends beyond simple solubility; researchers are exploring the creation of responsive materials that change their properties in response to external stimuli like pH, temperature, or light – a key area in smart coatings and drug delivery systems. Furthermore, the integration of solution chemistry with nanotechnology opens up exciting possibilities for creating novel composite materials and targeted therapies. Imagine nanoparticles dispersed within a polymer matrix, or drug delivery systems that release their payload only in response to specific biomarkers. These are not futuristic concepts, but active areas of research poised to revolutionize various fields.
However, alongside these exciting possibilities, challenges remain. Understanding the complex interplay of factors influencing solution behavior in real-world conditions – including temperature fluctuations, ionic strength variations, and the presence of impurities – is crucial for reliable application. Developing sustainable and cost-effective methods for synthesizing novel solvents and electrolytes is also a significant priority. Moreover, ensuring the safety and environmental impact of these materials is paramount.
Ultimately, the continued exploration of solution chemistry is not simply an academic pursuit; it’s a vital engine for innovation. From improving the efficiency of energy storage to developing more effective pharmaceuticals and creating environmentally friendly industrial processes, the ability to understand and manipulate the behavior of dissolved substances is becoming increasingly indispensable. As we navigate an increasingly complex world, the insights gleaned from the study of solutions will undoubtedly play a crucial role in shaping a more sustainable and technologically advanced future.