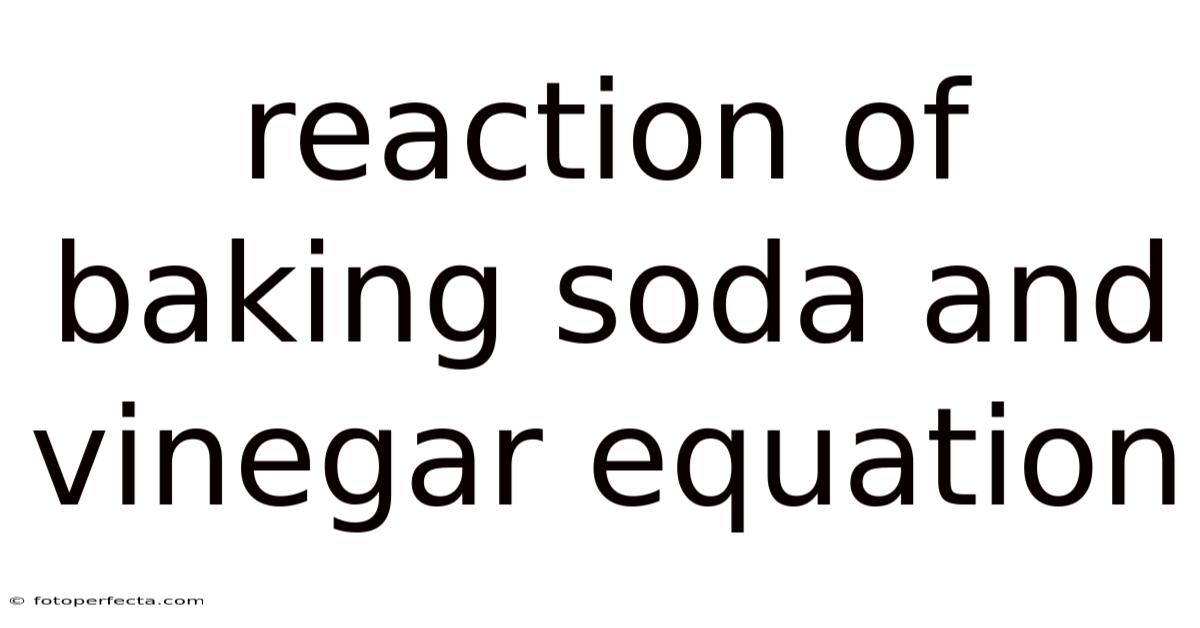The fizzing spectacle that greets you when sodium bicarbonate meets acetic acid is more than a kitchen trick—it’s a textbook demonstration of acid‑base chemistry that can be explained with a simple balanced equation. But this reaction, often called the baking soda and vinegar experiment, is a perfect example of how two common household substances combine to produce a gas, a salt, and water. By exploring the reaction equation, the underlying principles, and practical variations, you’ll gain a deeper appreciation for the science behind the bubbles and the versatility of this classic demonstration.
Introduction: Why the Reaction Matters
When you pour vinegar (an acid) into a container of baking soda (a base), the resulting effervescence is a visual proof that acids and bases react to form salt and water. This reaction is not only fun for kids but also a foundational concept in chemistry, illustrating neutralization, gas evolution, and the importance of balancing chemical equations. Understanding the equation helps you predict the products, control the reaction, and even design experiments that showcase other principles such as pH changes or catalyst effects.
The core reaction can be expressed as:
NaHCO₃ (s) + CH₃COOH (aq) → CO₂ (g) + H₂O (l) + CH₃COONa (aq)
Let’s break down each component and see how the math of the equation mirrors the chemistry of the fizz Worth keeping that in mind..
Step 1: Identifying the Reactants
| Reactant | Formula | Common Name | State |
|---|---|---|---|
| Sodium bicarbonate | NaHCO₃ | Baking soda | Solid (s) |
| Acetic acid | CH₃COOH | Vinegar (concentrated) | Aqueous (aq) |
In everyday use, vinegar is typically a 5 % solution of acetic acid in water. When we write the equation, we often simplify it to “acetic acid” because the water is a spectator that doesn’t participate directly in the chemical transformation Still holds up..
Step 2: Recognizing the Products
| Product | Formula | Common Name | State |
|---|---|---|---|
| Carbon dioxide | CO₂ | Gas | Gas (g) |
| Water | H₂O | Water | Liquid (l) |
| Sodium acetate | CH₃COONa | Salt | Aqueous (aq) |
Not obvious, but once you see it — you'll see it everywhere Easy to understand, harder to ignore..
The gas released is what creates the bubbling effect. Sodium acetate remains dissolved in the solution, giving the mixture a slightly salty taste. Water is formed as part of the neutralization process Not complicated — just consistent..
Step 3: Balancing the Equation
The unbalanced reaction:
NaHCO₃ + CH₃COOH → CO₂ + H₂O + CH₃COONa
To balance it, we count atoms on both sides:
- Sodium (Na): 1 on each side.
- Carbon (C): 1 in NaHCO₃, 2 in CH₃COOH (1 in CH₃COONa, 1 in CO₂). Balanced.
- Hydrogen (H): 1 in NaHCO₃, 4 in CH₃COOH (2 in H₂O, 2 in CH₃COONa). Balanced.
- Oxygen (O): 3 in NaHCO₃, 2 in CH₃COOH (2 in CO₂, 1 in H₂O, 2 in CH₃COONa). Balanced.
All atoms are already balanced, so the equation stands as written. This confirms that the reaction is a simple acid–base neutralization followed by a decomposition of the bicarbonate to release carbon dioxide.
Scientific Explanation: The Chemistry at Play
-
Acid–Base Neutralization
Acetic acid donates a proton (H⁺) to the bicarbonate ion (HCO₃⁻), forming a neutral molecule (CO₂) and water (H₂O). The reaction releases energy in the form of heat, albeit small enough to be felt as a mild warmth on the surface. -
Decomposition of Bicarbonate
Sodium bicarbonate is a weak base that, when protonated, breaks down into carbon dioxide and water. This decomposition is a classic example of a carbamate breakdown, often used in industrial applications to generate CO₂ on demand But it adds up.. -
Formation of Sodium Acetate
The remaining sodium ion (Na⁺) pairs with the acetate ion (CH₃COO⁻) from the acetic acid, forming sodium acetate, a soluble salt that stays in the liquid phase. -
Gas Evolution
The release of CO₂ gas is what creates the fizz. The gas bubbles rise to the surface, and the reaction can continue as long as reactants remain available.
Variations and Extensions
1. Changing Acid Strength
- Citric Acid: Replace vinegar with a citric acid solution (found in lemon juice). The reaction produces the same products but can be slower due to citric acid’s weaker acidity compared to acetic acid.
- Hydrochloric Acid: Using a dilute HCl solution (e.g., 1 % HCl) speeds up the reaction dramatically, producing a vigorous fizz and more CO₂.
2. Modifying Base Concentration
- Powdered Baking Soda vs. Liquid: Adding baking soda in liquid form (e.g., from a baking soda solution) can lead to a more controlled release of CO₂, useful in science projects that require a steady gas flow.
3. Temperature Effects
- Cold vs. Hot: Conducting the experiment at lower temperatures slows the reaction, allowing students to observe the gradual bubble formation. Higher temperatures increase the rate, producing a rapid, foamy eruption.
4. Measuring CO₂ Production
- Gas Collection: By placing a balloon over the reaction vessel, you can capture the CO₂ gas and measure its volume, providing a quantitative way to study stoichiometry.
5. pH Indicator Use
- Litmus Paper: Dip a strip of litmus paper in the reaction mixture before and after mixing. The color change from red to blue confirms the neutralization of the acidic solution by the base.
FAQ: Common Questions About the Reaction
| Question | Answer |
|---|---|
| **Does the reaction produce heat? | |
| **Is the reaction dangerous?That said, | |
| **Why does the reaction stop? That said, | |
| **What if I use too much vinegar? That's why ** | Yes, but the temperature change is minimal—usually a few degrees Celsius. That said, avoid ingesting the solution after the experiment. On the flip side, |
| **Can I reuse the leftover solution? On the flip side, both reactants are common household items, and the reaction is mild. ** | No. ** |
The official docs gloss over this. That's a mistake.
Conclusion: From Kitchen Counter to Classroom
The baking soda and vinegar reaction equation is a gateway into understanding acid–base chemistry, gas evolution, and stoichiometry. Which means by balancing the equation, we see that every atom is accounted for, reinforcing the law of conservation of mass. The experiment’s simplicity belies its educational power: it demonstrates key concepts in a way that is tangible, memorable, and safe Worth keeping that in mind..
Whether you’re a teacher preparing a lesson plan, a parent looking for a fun science activity, or a curious learner, this reaction offers a hands‑on way to witness the principles of chemistry in action. Grab a bottle of vinegar, a scoop of baking soda, and let the bubbles tell the story of how acids and bases meet, neutralize, and create a burst of carbon dioxide—proof that even the simplest household items can reveal the profound beauty of science.
Expanding the Experiment: Advanced Concepts
For those looking to delve deeper into the chemistry of the baking soda and vinegar reaction, there are several avenues to explore. Think about it: one could investigate the effect of different acids, such as citric acid or hydrochloric acid, on the rate of the reaction. This would introduce the concept of acid strength and its impact on reaction kinetics.
Another interesting extension is to study the role of catalysts. So adding a small amount of a catalyst, like manganese dioxide, can significantly speed up the reaction, allowing for a more controlled and rapid release of CO₂. This could lead to discussions about activation energy and how catalysts lower the energy barrier for a reaction to occur Surprisingly effective..
Real-World Applications
The principles demonstrated in this simple reaction have practical applications in various fields. That's why in environmental science, understanding gas evolution is crucial for monitoring carbon dioxide levels and developing strategies for carbon capture. In industry, the controlled release of gases is important for processes like fermentation and the production of plastics That alone is useful..
In the culinary world, the reaction between baking soda and acidic ingredients like vinegar is used to leaven baked goods, creating the light, airy texture that makes bread and cakes rise. This real-world application underscores the importance of chemistry in everyday life.
Conclusion: A Foundation for Future Exploration
The baking soda and vinegar reaction is more than just a fun kitchen experiment; it's a foundational experiment that can spark curiosity and provide a springboard for learning. By observing the reaction, students and enthusiasts alike can grasp fundamental concepts in chemistry, from stoichiometry to reaction kinetics, and see the relevance of these concepts in the real world And it works..
As we wrap up, remember that science is not just about knowing facts but about asking questions, experimenting, and discovering. The baking soda and vinegar reaction is a perfect example of how the simplest of experiments can lead to profound insights. So, whether you're a seasoned scientist or a curious beginner, dive into the world of chemistry with the baking soda and vinegar reaction, and let the bubbles lead the way It's one of those things that adds up. Which is the point..
You'll probably want to bookmark this section.